全固体電池(ASSB)の研究開発および工業化において、無機固体電解質(ISE)は中核となる重要な材料とみなされています。これらは酸化物系(例:LLZO)、硫化物系(例:Li3PS4、Li10GeP2S12)、またはハロゲン化物系のいずれに属するかにかかわらず、その物理的形態、粒度分布、および比表面積が電池の最終的な性能を直接決定します。. 超微粉砕(通常はマイクロメートル、あるいはナノメートルスケールまで)は、高性能固体電解質を実現するための必須の道筋です。新しい粉砕装置が登場しているにもかかわらず、従来のボールミル(遊星ボールミルやドラムボールミルを含む)は、研究および工業生産において依然として主流の選択肢となっています。これらは、低コスト、簡単な操作、および調整可能なエネルギー密度といった利点から好まれています。本稿では、従来のボールミルプロセスを用いて無機固体電解質を効率的に超微粉砕する方法を詳しく解説します。.
1. 無機固体電解質はなぜ「超微粉砕」が必要なのか?
プロセスの詳細に入る前に、粉砕の目的を理解しておく必要があります。固体電解質の場合、粒子サイズを小さくすることは単に「細かくする」ことだけではありません。以下の主要な問題を解決することが目的です。
- 界面インピーダンスの低減固体電池は「固体同士」の接触に依存しています。粒子サイズが小さいほど比表面積が大きくなります。正極活物質と混合されると、より密な接触ネットワークが形成されます。これにより、界面電荷移動インピーダンスが大幅に低減されます。.
- 電解質膜密度の向上プレス成形またはテープキャスティング工程において、超微細粉末は気孔が少ない。これにより密度が高くなり、リチウムデンドライトの侵入を効果的に防ぐことができる。.
- 反応速度の促進後続の熱処理が必要な材料の場合、超微細加工によって原子拡散距離が短縮されます。これにより、焼結温度を下げたり、反応時間を短縮したりすることが可能になります。.
2. 伝統的な物理的メカニズム ボールミル
ボールミル粉砕プロセスは、単に材料を「粉砕」するだけではありません。複雑な機械化学的作用が伴います。主な力は以下のとおりです。
- インパクトフォースボールは遠心力によって高いところから落下したり、対象物に衝突したりする。これにより瞬間的に大きな圧力が発生し、粒子が破壊される。.
- せん断力これは、ボール同士、およびボールとミル壁との間の相対的な滑りによって生じる研削効果です。.
- 摩擦充填率が高い場合、ボールが材料を圧縮することによって生じる摩擦が、研削効果に寄与する。.
無機固体電解質、特に柔らかいが変形しやすい脆い酸化物や硫化物の場合、せん断力と衝撃力のバランスを取ることが、超微粉砕を実現するための鍵となる。.
3. 効率的な粉砕のための主要プロセスパラメータの調整
従来のボールミルの効率を最大限に高めるには、以下の変数を正確に制御する必要があります。
3.1 ボール対粉末比(BPR)および充填速度
BPRとは、粉砕媒体(ボール)と材料(粉末)の質量比を指します。.
- 効率化に関する提案超微粉砕の場合、通常は高いBPR(例えば20:1または40:1)が使用されます。.
- 論理ボールの数が多いほど、単位時間あたりの材料への衝撃頻度が高くなります。しかし、比率が高すぎると容器内のスペースが不足し、ボールの動きが妨げられる可能性があります。.
3.2 粉砕媒体のサイズ構成
“「粒度選別」は、超微粉砕の真髄である。.
- 成形用の大きなボール大きな球体は運動エネルギーが高い。それらは大きな塊状物質の初期破壊を引き起こす原因となる。.
- 微粉砕用小型ボール粒子がマイクロメートルレベルまで縮小すると、大きな球体間の隙間が広くなりすぎて、材料が「すり抜けてしまう」。この段階では、最終的なナノメートルスケールの格子構造を形成するために、接触点を増やすために多数の小さな球体(例えば、0.1mm~0.5mm)が必要となる。.
- 段階的ボール法直径10mmから1mm以下まで、段階的に異なる直径の組み合わせを使用することをお勧めします。.
3.3 回転速度の最適化
速ければ良いというわけではない。.
- クリティカルスピード回転速度が速すぎると、遠心力によってボールが容器の壁に押し付けられ、落下しなくなるため、粉砕効率がゼロになってしまう。.
- 効率ゾーン通常は臨界速度の70%から85%に設定されます。この範囲では、ボールが「連鎖運動」を起こし、最大の衝撃エネルギーを生み出します。.
4. 乾式粉砕と湿式粉砕:どちらを選ぶべきか?

これは、超微粉砕を実施する際に最もよく直面する選択肢です。.
乾式粉砕
- 利点シンプルなプロセス。後処理としての溶媒除去は不要。溶媒による化学的劣化のリスクなし。.
- デメリット: 重度の凝集. 粉末が一定の細かさに達すると、分子間力(ファンデルワールス力)によって粒子が再び結合して塊を形成します。これにより、粉末がそれ以上細かくならないボトルネックが生じます。.
- 応用: 初期粉砕、またはあらゆる溶剤に極めて敏感な材料。.
ウェットミリング
- 利点液体媒体は粉末を効果的に分散させ、凝集を防ぎます。液体は粒子表面のエネルギーを低下させることで「粉砕助剤」として機能します。粉砕効率は乾式粉砕に比べて数倍高くなります。.
- 要点: 溶媒の選択.
- のために 硫化物電解質, 無水非極性溶媒(例えば、ヘプタン、トルエン、キシレン)を使用しなければなりません。そうしないと、激しい加水分解が起こります。.
- のために 酸化物 (例えばLLZOの場合)、プロトン交換反応(Li+/H+交換)を防ぐ必要がある。一般的には無水イソプロパノールまたはエタノールが用いられる。.
5.「効率性のボトルネック」を克服するための高度なテクニック“
実際には、適切なパラメータを設定しても、研削効率が頭打ちになることがあります。これを打破するためのいくつかの方法を以下に示します。
5.1 粉砕助剤の添加
少量の界面活性剤や特定の有機分子を添加すると効果的です。これらの分子は粒子の亀裂表面に吸着し、亀裂の修復を防ぎます。また、粒子間の静電引力も低減します。これは特に乾式粉砕において有効です。.
5.2 エネルギー密度管理:断続粉砕
連続的な高速研削は、かなりの熱を発生させる。硫化物系固体電解質の場合、熱によって材料が軟化したり、相転移(結晶状態からガラス状態への変化)を起こしたりする可能性がある。.
- 戦略「10分間の研削後、5分間の休憩」といったサイクルを使用してください。これに水冷システムを組み合わせます。これにより、材料は低温状態でも応力がかかり続け、その脆性を利用して迅速に破壊されます。.
5.3 材料のマッチング:汚染の回避
超微粉砕の過程では、ボールと容器の摩耗は避けられない。.
- 原理研削媒体の硬度は、被削材の硬度よりも高くなければならない。.
- トップチョイス: ジルコニア(ZrO₂). ジルコニアは極めて高い硬度と靭性を有しています。さらに、微量のジルコニアの摩耗は、ほとんどのリチウム電池電解質の電気化学的性能に比較的悪影響を与えません。.
6. さまざまな電解質システムに対する具体的な最適化アドバイス
6.1 酸化物系(例:LLZO、LATP)
酸化物は非常に硬度が高く、研磨が難しい。.
- 解決2段階の「乾式粉砕後湿式粉砕」法が推奨されます。まず、大きなボールを用いて乾式粉砕を行い、約10μmの粒径にします。次に、溶媒を加え、小さなボールを用いて湿式粉砕を長時間行い、500nm以下の粒径にします。.
6.2 硫化物系(例:Li2S-P2S5)
硫化物は硬度が低いが、酸化や凝集を起こしやすい。.
- 解決: グローブボックスのフル操作 (不活性ガス雰囲気下での)粉砕は必須です。湿式粉砕法を用いなければなりません。局所的な過熱によるイオン伝導率の低下を防ぐため、粉砕温度は厳密に管理する必要があります。.
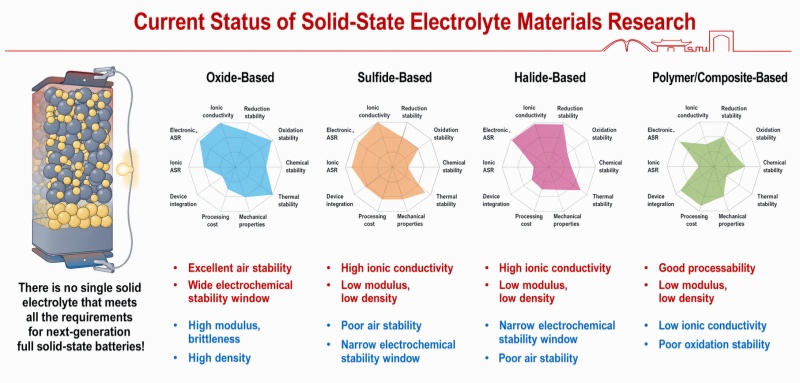
7. まとめ アウトルック
従来のボールミルを用いて無機固体電解質を効率的に超微粉砕するには、バランス感覚が求められる。運動エネルギーの消費と熱効果、破砕と凝集、そして微粉砕度と純度のバランスを取る必要がある。.
高いボール対粉末比、多段階ボール粒度選別、最適化された速度、そして科学的な湿式媒体の選定により、従来型のボールミルは高品質の超微粉末を製造する能力を十分に備えています。これらの粉末は、実験室規模はもちろん、パイロットスケール生産の要件も満たしています。.
しかし、工業化が進むにつれて、より狭い粒度分布と連続生産が求められるようになるため、ボールミル法はビーズミル法やジェットミル法といった技術とますます統合され、補完し合うようになるだろう。.
固体電池の研究開発に携わるすべてのエンジニアにとって、ボールミルの「特性」を習得することは極めて重要です。継続的な実験を通して、その「最適なパラメータ」を見つけることが、高性能固体電池への扉を開く鍵となります。.

読んでいただきありがとうございます。この記事がお役に立てれば幸いです。ぜひ下のコメント欄にご意見をお寄せください。また、ご質問等ございましたら、Zeldaのオンラインカスタマーサポートまでお問い合わせください。
— 投稿者 エミリー・チェン
