In recent years, lithium-ion batteries have been widely used in power and energy storage markets. As a result, lithium resources have become increasingly scarce. Sodium-ion batteries operate on a similar principle and show comparable performance to lithium-ion batteries. However, compared with lithium-ion batteries, sodium-ion batteries have several clear advantages:
- Abundant raw material reserves
- Wide distribution
- Low cost
- Environmental friendliness
- Compatibility with existing lithium-ion battery production equipment
They also offer good power performance, wide temperature adaptability, high safety, and no over-discharge problems. Therefore, sodium-ion batteries are widely regarded as an important alternative technology for large-scale energy storage.
Because the ionic radius of Na⁺ is significantly larger than that of Li⁺, cathode materials suitable for lithium-ion batteries are not necessarily suitable for sodium-ion batteries. Developing cathode materials with larger ion transport channels has thus become a key factor in advancing sodium-ion battery technology.
The main cathode materials for sodium-ion batteries include three categories:
- Transition metal oxides
- Polyanionic compounds
- Prussian Blue analogues (PBAs)
Among them, Prussian Blue analogues (PBAs) have attracted extensive attention. This is due to their unique open framework and three-dimensional large-channel structure. These features provide abundant sodium storage sites and smooth ion insertion/extraction pathways. As a result, PBAs are particularly suitable for accommodating and storing the larger Na⁺ ions.

Prussian Blue and Prussian Blue Analogue Cathode Materials
Prussian Blue (PB) is an iron hexacyanoferrate coordination compound, represented as Fe³⁺₄[Fe²⁺(CN)₆]₃⁻ or Fe²⁺[Fe³⁺(CN)₆]₃⁻, abbreviated as Fe-HCF. Without altering the overall PB framework structure, replacing Fe with other metal elements results in a class of new compounds generally referred to as Prussian Blue analogues (PBAs).
The general structural formula of PBAs is:
NaxM[Fe(CN)₆]₁–y·□y·zH₂O
where M represents transition metal elements such as Fe, Co, Ni, or Mn; □ denotes Fe(CN)₆ vacancies; 0 < x < 2; and 0 < y < 1.
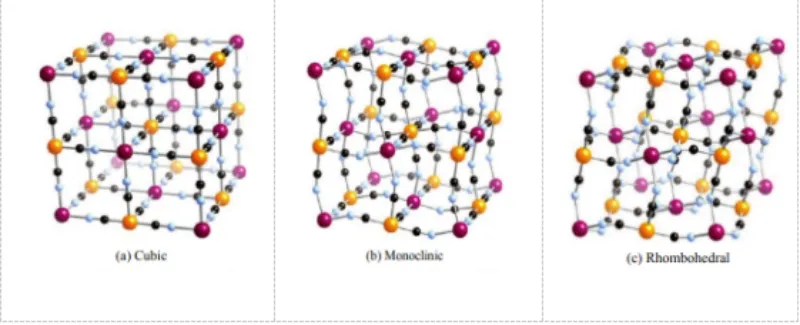
The crystal structure of PBAs features a unique three-dimensional open framework. It forms through coordination between transition metal M and Fe with the N and C atoms of CN⁻, respectively. Na⁺ ions occupy the interstitial sites, while crystalline water exists on the surface and inside the crystals.
PBAs generally exhibit a face-centered cubic structure. However, differences in preparation processes lead to variations in Na⁺ and crystalline water content. These variations may distort the crystal structure into monoclinic or rhombohedral systems. When the transition metal M connected to the N atom of CN⁻ changes, the electrochemical performance of the material also varies.
If M is electrochemically inactive, such as Ni, Zn, or Cu, only one Na⁺ can reversibly insert and extract during cycling. The theoretical capacity is about 85 mAh/g. If M is electrochemically active, such as Fe, Co, or Mn, two Na⁺ ions can participate in reversible reactions. The theoretical capacity can reach approximately 170 mAh/g.
Prussian Blue analogues exhibit many advantages as cathode materials for sodium-ion batteries, mainly including:
- A large three-dimensional channel structure and abundant storage sites, facilitating Na⁺ migration and storage.
- A rigid framework with minimal volume change during Na⁺ insertion/extraction, leading to good cycling stability.
- Low migration energy barriers for Na⁺, enabling rapid ion transport and improving power density.
- Certain modified materials possess two redox electron pairs, providing high specific capacity.
- Simple synthesis process and low cost, suitable for large-scale production.
- Environmentally friendly, non-toxic, and pollution-free.
However, PBAs often contain considerable crystalline water and Fe(CN)₆ structural defects after synthesis. Lattice water can occupy sodium storage sites and diffusion channels, reducing Na content and slowing ion migration. This weakens electrochemical performance. In addition, coordinated water and Fe(CN)₆ vacancies in the MHCF framework may trigger structural collapse during cycling, reducing stability. Therefore, researchers continue optimizing synthesis routes and applying modification strategies to obtain PBAs with low water content, fewer defects, high crystallinity, and improved electrochemical performance.
Preparation Methods of Prussian Blue Analogue Cathode Materials
Currently, the main synthesis methods for PBAs used in sodium-ion batteries can be classified into liquid-phase and solid-phase methods. Liquid-phase methods mainly include co-precipitation and hydrothermal methods, while solid-phase methods primarily involve mechanical ball milling.
Among them, the co-precipitation method is simple to operate, offers good process controllability, and enables large-scale continuous production. It has significant industrial application potential and is currently the primary method adopted by universities, research institutes, and industrial companies for both performance research and mass production of PBA cathode materials.
3.1 Co-precipitation Method
The co-precipitation method is the earliest and most commonly used approach for synthesizing PBAs. Early preparation mainly employed rapid precipitation. Later studies revealed that the crystallinity of PBAs directly affects their electrochemical performance. To improve crystallinity, chelating-agent-assisted slow co-precipitation methods were introduced.
Common chelating agents include trisodium citrate, sodium oxalate, sodium pyrophosphate, and ethylenediaminetetraacetic acid (EDTA).
Besides crystallinity, crystalline water content, structural defects, and Na content in MHCF structures also significantly affect electrochemical performance. To reduce crystalline water content, researchers optimize drying methods, introduce additives, adjust solvent formulations, and refine reaction time and temperature.
Although slow co-precipitation is time-consuming, it allows easy process adjustment and enables the synthesis of high-crystallinity, low-water-content, low-defect, high-sodium-content PBAs with excellent electrochemical performance.
3.2 Hydrothermal Method
In addition to co-precipitation, the hydrothermal method has also been successfully applied to synthesize PBAs (especially FeHCF). Liu et al. used different concentrations of HCl in a hydrothermal process to synthesize FeHCF with varying morphologies.
When 1 mL of HCl was added, cubic FeHCF particles were obtained. With 2 mL of HCl, the particle surface became somewhat rough. When increased to 3 mL, the morphology transformed into spherical particles. The cubic FeHCF exhibited the best electrochemical performance, delivering a capacity of 107 mAh/g at 0.2 A/g with 74% capacity retention after 500 cycles. Even at a high current density of 5 A/g, it maintained a capacity of 82 mAh/g.
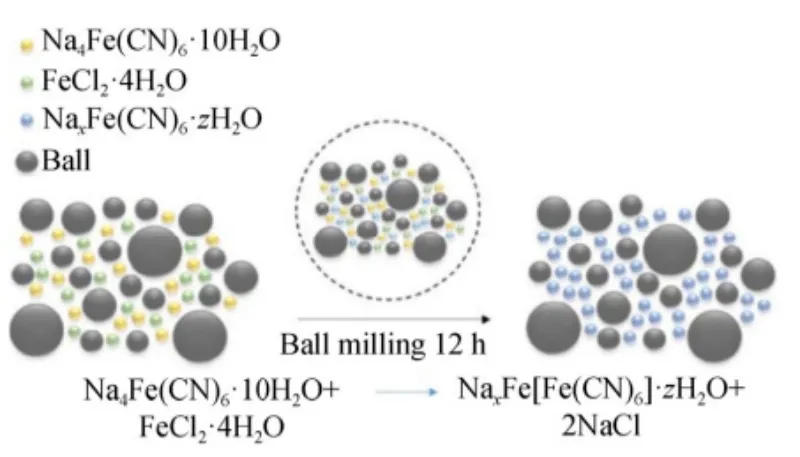
3.3 Ball Mill Method

The ball milling method uses mechanical vibration and impact to reduce large particles into nanoscale powders. It is suitable for synthesizing materials with low interstitial water content. The process is simple and can reduce crystalline water and particle size.
However, primary particles obtained by this method tend to agglomerate, solid–solid reactions may be incomplete, and impurities can be introduced. Additionally, materials synthesized by ball milling are currently relatively limited, mainly focusing on FeHCF.
Modification of Prussian Blue Analogue Cathode Materials
Besides optimizing synthesis processes, PBAs can be modified through composite formation with other materials or via ion doping.
4.1 Composite Modification
PB and PBAs can be combined with other materials (such as carbon materials, organic polymers, and graphene) to obtain cathode composites with improved conductivity, faster ion transport, enhanced rate performance, and longer cycle life.
Composite with Carbon Materials
Carbon materials are widely used not only as active electrode materials but also as conductive matrices due to their high electronic conductivity. They enhance conductivity, suppress particle aggregation, improve structural stability during cycling, and serve as buffering matrices to alleviate electrode expansion during Na⁺ insertion/extraction. Thus, forming composite electrodes with carbon materials is an effective strategy to improve electrochemical performance.
Composite with Organic Conductive Polymers
Organic conductive polymers (such as polyaniline, polypyrrole, and poly(3,4-ethylenedioxythiophene)) offer advantages including high energy storage capability, low cost, tunable physicochemical properties, and good environmental stability. Compositing PBAs with these polymers is an effective method to enhance electrochemical performance.
Composite with Graphene
Most PB and PBA materials suffer from poor conductivity and structural instability. Graphene, with its excellent electrochemical properties and large specific surface area, abundant edge sites, and defects, facilitates rapid sodium-ion transport and significantly improves conductivity when combined with PB/PBAs.
4.2 Doping Modification
Doping is another common modification strategy. Appropriate doping can reduce band gaps and migration energy barriers, thereby enhancing electron and Na⁺ mobility.
Doping with larger-radius metal ions can expand lattice parameters, increase sodium storage sites, and widen Na⁺ diffusion channels. Introducing electrochemically active metal ions can increase capacity, while incorporating electrochemically inactive metal ions can act as structural pillars to improve cycling stability.
For PBAs, doping is typically performed at the transition metal site coordinated with nitrogen. Since NiHCF exhibits excellent cycling stability, Ni doping is often used to modify FeHCF, MnHCF, and CoHCF cathode materials.

Conclusion
Prussian Blue analogue cathode materials demonstrate excellent sodium storage performance due to their unique open framework structure, abundant sodium storage sites, and large sodium-ion migration channels. However, during synthesis, crystalline water and Fe(CN)₆ vacancies are easily formed, significantly affecting electrochemical performance.
Although optimizing synthesis processes, forming composites with other materials, and applying ion doping can improve sodium storage performance, further research is still required to achieve large-scale industrial production.

“Thanks for reading. I hope my article helps. Please leave a comment down below. You may also contact Zelda online customer representative for any further inquiries.”
— Posted by Emily Chen
